Enantioselective Epoxypyrrolidines via a Tandem Cycloaddition/Autoxidation in Air and Mechanistic Studies
BY: Kaixiu Luo, Yongqiang Zhao, Jiawei Zhang, Jia He, Rong Huang, Shengjiao Yan*, Jun Lin*, and Yi Jin*
Key Laboratory of Medicinal Chemistry for Natural Resource, Ministry of Education and Yunnan Province, School of Chemical Science and Technology,
Yunnan
University
,
Kunming
650091
,
P. R. China
Organic Letter, 2019, 21 (2), pp 423–427
DOI: 10.1021/acs.orglett.8b03605
Publication Date (Web): December 27, 2018
Document Type:Article
Abstract
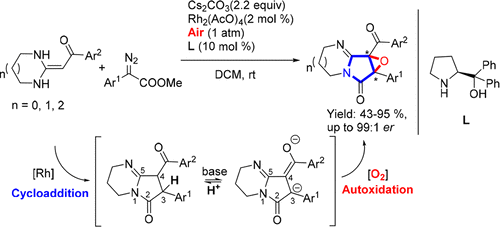
A tandem cycloaddition/autoxidation reaction between heterocyclic ketene aminals and diazoester in air is described for the enantioselective preparation of epoxypyrrolidines. Notably, the results of mechanistic studies suggest that epoxide was oxidized from an sp3 C–C single bond, which is of mechanistic and practical interest as this protocol may be suitable for constructing other bioactive heterocyclic epoxides.
全文链接:https://pubs.acs.org/doi/10.1021/acs.orglett.8b03605