Total synthesis of (−)-vindoline
By: Wen Chen, Hongchang Tian, Wenyun Tan, Xiaotong Liu, Xiaodong Yang, Hongbin Zhang
Key Laboratory of Medicinal Chemistry for Natural Resource, Ministry of Education and Yunnan Province, School of Chemical Science and Technology, Yunnan University, Kunming, Yunnan, 650091, People's Republic of China
Tetrahedron
Available online 28 November 2018
Abstract
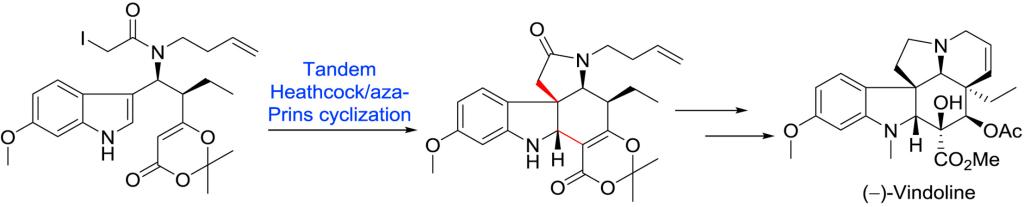
In this full paper, a stereocontrolled strategy for the total synthesis of (−)-vindoline is described. This synthetic route features: 1) rapid construction of the stereochemical center at C19 through a highly diastereoselective vinylogous Mannich addition; 2) tandem Heathcock/aza-Prins cyclization to install rings C and E in vindoline; 3) oxidative transformation of β-ketoester to enone; 4) stereoselective inversion of C4 stereochemistry with triphenylphosphine and carbon tetrabromide followed by Brønsted acid.
全文链接:https://www.sciencedirect.com/science/article/pii/S0040402018314017